The WHO approves monkeypox vaccine experiment on adults and children in Africa, despite knowing it causes cardiac events
09/19/2024 / By Lance D Johnson

As the World Health Organization (WHO) loses standing around the globe for deceiving populations with lockdowns, experimental vaccines and unlawful mandates, the corrupt behemoth has decided now to go all-in with a new experiment on African adults and children.
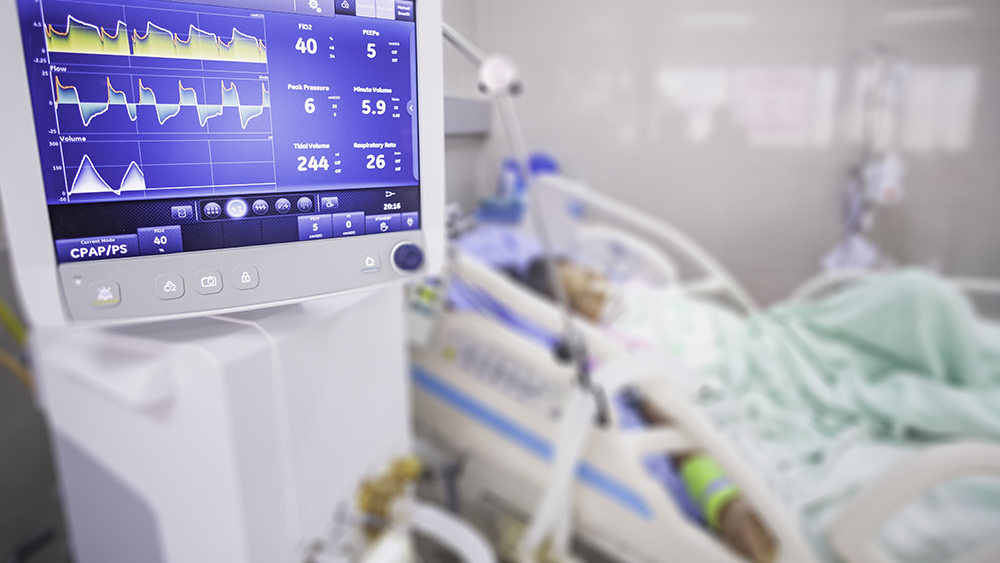
The WHO recently approved Bavarian Nordic’s MVA-BN vaccine for monkeypox in adults, children and pregnant women, even though the vaccine is not licensed for individuals under 18 years of age and causes cardiac events in 2% of adult recipients. This decision has drawn widespread condemnation from public health experts and medical freedom groups who argue that the vaccine’s hasty approval reflects a dangerous and unethical approach to a manufactured crisis, with no medical precedent to necessitate its widespread use in populations.
WHO unleashes the monkeypox scamdemic on Africa
After multiple nations rejected the WHO’s pandemic treaty in the summer of 2024, and with a growing list of American governors rejecting the WHO’s authority; the inter-governmental agency must act quickly and boldly, as they seek to remain relevant in a world of manufactured crises. What better way to remain relevant as a global terror organization than to sell Africa on a new monkeypox scamdemic? WHO will allow the nations of the world to unleash a new wave of vaccine experiments that will inevitably cripple and maim victims with new cardiac events.
The WHO’s endorsement of the “Modified Vaccinia Ankara-Bavarian Nordic (MVA-BN) vaccine, also known in the U.S. as Jynneos, comes despite significant concerns about its safety profile, particularly in pediatric populations and pregnant women. The vaccine, primarily designed to combat smallpox and monkeypox, has been authorized for use in outbreak settings where the benefits are purported to outweigh the risks. This approval is expected to facilitate the procurement of the vaccine by governments and international organizations such as GAVI and UNICEF, which facilitate mass vaccination of children living in the world’s poorest countries.
Brian Hooker, Chief Scientific Officer at Children’s Health Defense, labeled the WHO’s decision as “a train wreck in the making,” emphasizing that the safety and efficacy of the vaccine in children and pregnant women have not been adequately tested. “The safety profile is abysmal in adults, with up to 2.1% experiencing serious cardiac events in clinical trials. There is no reliable data on its effects on children or pregnant women, which raises serious concerns about potential harm,” Hooker said.
Dr. David Bell, a former WHO medical officer and current public health physician, echoed these concerns, criticizing the WHO for focusing on monkeypox vaccines at the expense of addressing more pressing health issues in Africa. He highlighted that approximately 40,000 children have died from malaria alone in the Democratic Republic of Congo (DRC) this year, with similar numbers suffering from malnutrition, tuberculosis and HIV/AIDS.
“We have become much better at detecting much rarer diseases such as mpox, and addressing these is certainly more lucrative for the growing industry feeding off the WHO’s misinformation regarding rapidly rising pandemic risk,” he said.
Bell accused the WHO of prioritizing the needs of the pharmaceutical industry over addressing these critical public health issues, suggesting that the organization’s approach reflects a return to colonialist-era practices rather than evidence-based public health strategies.
WHO approves monkeypox vaccines without clinical trials
The WHO’s approval process for the MVA-BN vaccine involved a review by its Strategic Advisory Group of Experts on Immunization (SAGE), which noted the absence of specific clinical trials for children. The situation is compounded by the fact that the vaccine’s use in the U.S. has been limited to high-risk individuals under the age of 18, with additional scrutiny recommended for very young children and pregnant women. This raises questions about the appropriateness of extending its use to broader populations in Africa without similar safeguards.
The vaccine’s safety profile is not good, with recipients reporting pain at site of injection (85% of recipients); redness, swelling, itching, and induration at site of injection (40–60%); fatigue, muscle pain, and headaches (20–40%); nausea (17%), and chills (10%). There were also vaccine-induced fevers in 2% of recipients and cardiac events in around 2.1% of recipients.
A WHO press release said the vaccine can be given to adults over 18 in a two dose series, spaced out four weeks apart. They also fast-tracked single dose vaccines “in supply constrained outbreak situations.” WHO admitted they need more data on the vaccine’s safety and efficacy in these situations, but they approved it anyway. WHO admitted that “while MVA-BN is currently not licensed for persons under 18 years of age” it can be used “off-label in infants, children and adolescents, and in pregnant and immunocompromised people.”
The WHO’s recent decision has not only sparked controversy but also calls for a reevaluation of how public health interventions are prioritized and implemented, especially in regions with pressing health needs and limited resources.
Sources include:
Submit a correction >>
Tagged Under:
adverse events, big government, cardiac events, Censored Science, chills, experiments, GAVI, global emergencies, Globalism, headaches, medical ethics, monkeypox, poisoning, scam, STDs, UNICEF, vaccine damage, vaccine safety, vaccine wars, vaccines, WHO
This article may contain statements that reflect the opinion of the author