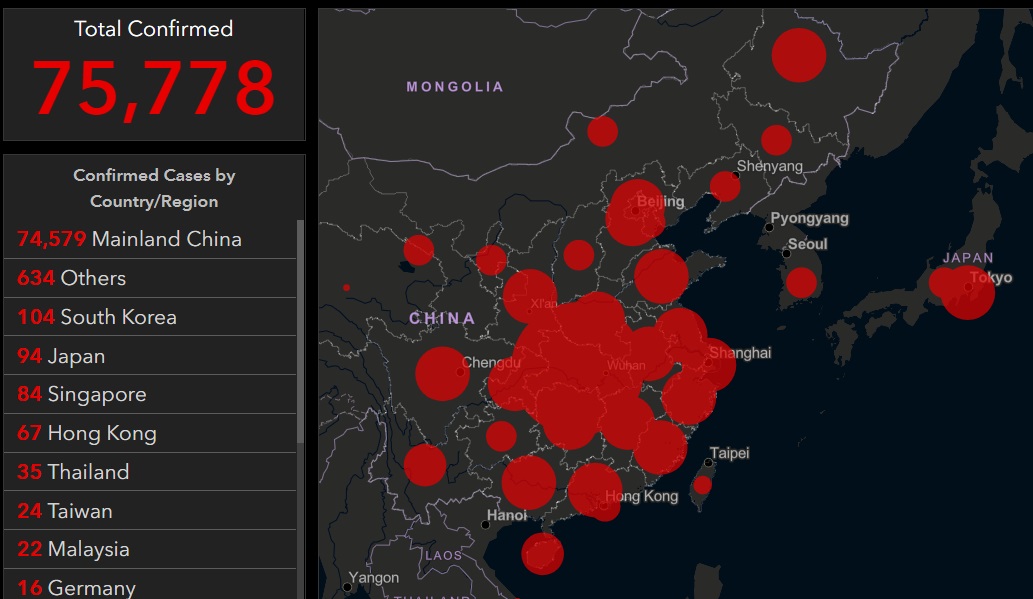
Researchers discover how to deactivate influenza protein that turns off innate immunity
02/20/2020 / By Evangelyn Rodriguez
Research on the influenza virus has come a long way. One of the most significant discoveries about the virus is the role of Non-Structural Protein 1 (NS1) in influenza virus A infection and pathogenesis.
According to earlier studies, NS1 plays a critical role by antagonizing the host cell’s anti-viral response. But while scientists are familiar with the many ways this protein performs its function, not all of the mechanisms involved in it are clear.
In a recent study published in the Journal of Biological Chemistry, researchers from the University of Alabama (UA) explored the interactions between influenza A NS1 and a protein found in the cytoplasm of human cells called retinoic acid-inducible gene I (RIG-I). RIG-I is an important receptor that recognizes and responds to viral nucleic acids by triggering the cell’s innate anti-viral defense. The researchers found that a specific mutation in NS1 reduces the influenza A virus’ ability to prevent the activation of RIG-I, which is needed in order for cells to mount an appropriate immune response.
How influenza A NS1 shuts down innate immunity
According to UA researcher Chad Petit, NS1 is responsible for modulating the virulence of the influenza virus. Previous studies have found that its two functional domains — an RNA-binding domain (NS1RBD) and an effector domain (NS1ED) — antagonize the cell’s type I interferon (IFN)-mediated immune response to viral infections. But when a conserved portion of arginine and lysine residues in NS1RBD is mutated, the influenza virus loses its ability to block IFN production upon infection. IFN provides one of the first lines of host defense against virus infection.
The conserved portion in NS1RBD is involved in the interaction between NS1 and the type I IFN response activator, RIG-I. As the cell’s innate virus sensor, RIG-I binds to viral RNAs. This causes its structure to change and leaves its two functional domains, CARD1 and CARD2, exposed. In the presence of other viruses, CARD2 normally undergoes a series of reactions that eventually drives type I IFN production. However, the binding of influenza NS1RBD’s conserved portion to RIG-I suppresses RIG-I function, inhibiting the production of IFN and the activation of the cell’s anti-viral response.
A strain-specific mutation in the NS1 protein negates its function
Back in 2015, Petit, together with researchers from the University of Wisconsin-Madison, investigated the strain-specific interactions between viral and host proteins that dictate virulence. They noticed a difference between the severity of the H1N1 virus that caused the 1918 Spanish influenza pandemic and the 2009 H1N1 virus that caused the Swine Flu pandemic. Although both showed high transmissibility, the more recent strain of H1N1 displayed a much lower virulence than the old one.
When the researchers compared the 1918 H1N1 virus with a different influenza strain — the Udorn H3N2 virus — they found that the NS1RBD of the 1918 H1N1 virus directly interacts with CARD2, while the Udorn NS1RBD does not. This suggests that the interaction between NS1 and RIG-I is influenced by the strain of the virus. The researchers also identified several structural differences between the two NS1RBDs, which may influence NS1 binding to RIG-I as well.
In their latest study, Petit and his team used reverse genetics to engineer the mutation they found in the Udorn NS1RBD into the 1934 Puerto Rico influenza virus. They aimed to further understand the strain-specific interaction between NS1 and RIG-I and its biological effects. The researchers reported that, unlike the original NS1, the mutant NS1 protein did not antagonize RIG-I signaling. The mutation in its NS1RBD reduced the protein’s ability to bind to RIG-I, which led to the activation of cellular innate immunity. (Related: A natural cure for influenza: Study proves elderberries are an effective antiviral.)
According to Petit, comparisons of different NS1 sequences suggest that mutations in the NS1RBD of different strains may be related to species-specific adaptation.
“Taken together, the work presented in this study stresses the importance of how strain-specific polymorphisms in NS1 can affect its ability to antagonize the host cellular immune response in ways that are yet to be appreciated,” Petit said.
Sources include:
Tagged Under: anti-viral, breakthrough, discoveries, H1N1, immune system, immunity, infections, Influenza A, influenza strains, research, superbugs, viral infections, viral protein, virulence, Viruses